About Qartemi
Qartemi (varnimcabtagene autoleucel) is a groundbreaking immunotherapy for Relapsed / Refractory B Cell Non-Hodgkin Lymphoma. It is an autologous second-generation CAR T-cell product.
Qartemi consists of 4-1BB co-stimulatory domain and a non-FMC63 based murine single chain variable fragment targeting CD19 (A3B1 binder), manufactured in India by Immuneel Therapeutics.
Mechanism of Action
CAR T-cell therapy is a novel treatment to fight some cancers. It involves using body’s own immune system to treat your cancer.
The body has different types of blood cells, such as white blood cells. Lymphocytes are a part of white blood cells, which are an important part of your immune system that fights infections and cancers. These are broadly of two types – B-cells and T-cells.
T-cell lymphocytes are an integral part of your immune system and play an important role in fighting cancer. T-cells recognize abnormal cells, such as cancer cells, using proteins on their surface called receptors. They bind to proteins found on cancer cells and destroy them. Sometimes, cancer cells can survive by disguising themselves from being recognized by T-cells.
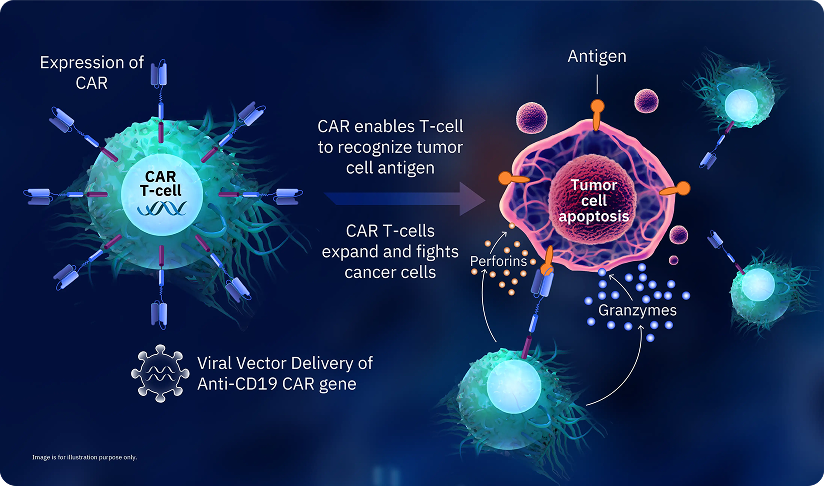
For CAR T-cell production, T-cells from your blood are collected through a process called Apheresis. Scientists in the laboratory then change the DNA of T-cells to make them produce a new type of receptor, called a Chimeric Antigen Receptor (CAR), they become CAR T-cells. These CAR T-cells are more efficient at binding to and removing cancer cells. They also send out signals that attract other immune cells and cause them to rapidly reproduce near the cancer cells. This increases the chance of eliminating cancer cells from the body. Thus, CAR T-cells can find and destroy cancer cells more effectively than ordinary T-cells.
CD-19 CAR T-cell therapy has been a standard of care for Relapsed/Refractory B-cell Malignancies in the West since 2017. Based on the recently published evidence, CD-19 targeted CAR T-cell therapy is now the preferred second line therapy for some sub types of B-cell Malignancies.
Indications and Dosage
Efficacy
Safety
cytokine release syndrome (CRS)
G3 + 0%
Overall 75% (n = 9/12)
Frequently asked questions
Before reaching out for support, we recommend checking our comprehensive Frequently Asked Questions (FAQ) section. In case your queries are not answered, please contact us.
check faq’s





